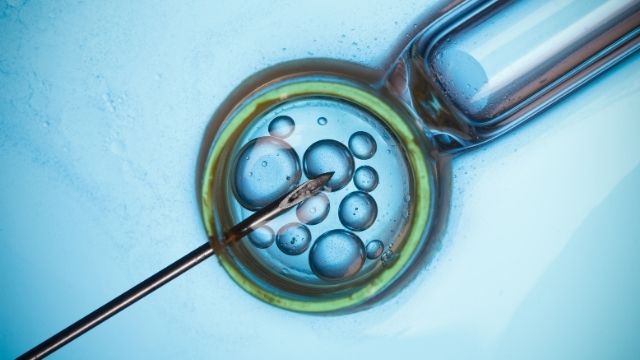
Navigating India’s Assisted Reproductive Technology (ART) Act: Laws, Provisions, and Considerations in Light of Sidhu Moosewala’s Parents’ IVF Pregnancy
Related Articles
Congress Supports Vijay for Tamil Nadu Government: तमिलनाडु में विजय की TVK को ‘हाथ’ का मिला साथ, लेकिन कांग्रेस ने रख दी एक शर्त!
Congress Supports Vijay for Tamil Nadu Government: तमिलनाडु में हाल ही में संपन्न चुनावों में तमिलगा वेत्री कड़गम (TVK) ने महत्वपूर्ण सफलता प्राप्त की...
Vijay Stakes Claim to Form Tamil Nadu Government with Congress Support?
Actor and politician Vijay has officially claimed a stake in forming the Tamil Nadu government following a meeting with Governor Rajendra Arlekar on May...
Indian Surgeon Performs Remote Robotic Surgery From Australia
An Indian bariatric surgeon, Dr Mohit Bhandari, successfully conducted a surgery from Perth, Australia, on a patient located in Indore, India, a distance of...