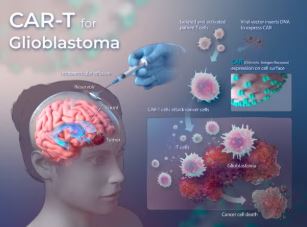
New CAR-T Cell Therapy Shows Promise in Shrinking Brain Cancer in Early Trial
Related Articles
Indian Navy Submarine Takshak: समुद्र में दुश्मन का काल बनेगा ‘तक्षक’, होगा साइलेंट किलर, दुश्मन को नहीं लगेगी भनक
ताकतवर टॉरपीडो का आ रहा है आगाज
Indian Navy Submarine Takshak: भारत अपनी समुद्री शक्ति को बढ़ाने के लिए एक नया कदम उठाने जा रहा...
‘I Multiplied Myself’: Taboola CEO Shares His Radical AI Productivity Hack
Adam Singolda, the CEO and Founder of Taboola, has recently shared insights on how his morning routine has been transformed by the use of...
Dispute Over Rs 50,000 Unveils Extensive Kidney Racket in Kanpur
A payment dispute involving Rs 50,000 has led to the uncovering of a suspected multi-crore illegal kidney transplant operation in Kanpur. Authorities have revealed...