Keytruda, known scientifically as pembrolizumab, is a leading immunotherapy drug that has established itself as a crucial option for treating numerous hard-to-manage cancers. Despite its recent approval by the US Food and Drug Administration (FDA) for a particular type of ovarian cancer, the majority of Indian cancer patients are unable to access this potentially life-saving treatment. The disparity in availability highlights significant economic barriers rather than scientific limitations.
Each year, over 1.5 million new cancer cases are reported in India, with more than 900,000 fatalities linked to various cancers. Yet, fewer than three per cent of patients can currently access novel immunotherapy treatments like Keytruda, which have been shown to enhance survival rates and improve quality of life.
Dr K M Gopakumar, a legal scholar associated with Third World Network, stated that the existing high costs are a major hurdle. The price of pembrolizumab could remain prohibitive for another two years, largely attributed to the drug’s ongoing patent protection.
Significance of Pembrolizumab in Cancer Treatment
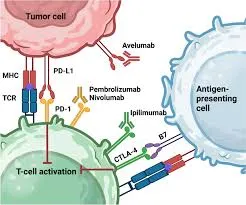
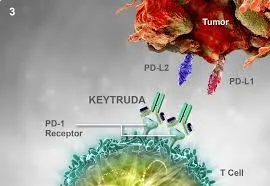
Since its initial approval in 2014, pembrolizumab has become integral to the care of various malignancies, including lung, skin, bladder, and kidney cancers. Its inclusion in the World Health Organisation’s Model List of Essential Medicines in September 2025 further underscores its status as a vital therapeutic option. The drug operates by enhancing the immune response against cancer cells, marking a significant shift from traditional treatment methods like chemotherapy.
The global demand for pembrolizumab reflects its effectiveness, making it the world’s best-selling cancer therapy, with annual sales in the tens of billions of dollars. In wealthier nations, it has significantly altered expectations regarding cancer treatment outcomes.
However, for many patients in India, accessing this treatment remains a distant goal. A complete course of pembrolizumab can reach up to Rs 50 lakh, translating to approximately Rs 1.2 crore per year for ongoing treatment. These figures place the therapy beyond the financial reach of the majority of Indian households, which typically rely on out-of-pocket healthcare expenditures.